Plamen Atanassov’s group investigates several subjects of critical importance in the electrochemical engineering field, mainly related to fuel cell technology and synthesis of value added products through alcohol oxidation or CO2 and N2 reduction reac-tions (CO2RR and NRR, respectively). Specifically, the main topics can be described as follow (i) M, N-doped carbonaceous electrocatalysts for the oxygen reduction reaction (ORR, i.e. the cathodic reaction of a proton exchange (or alkaline) membrane fuel cell – PEMFC), the CO2RR and NRR; (ii) Pt-based electrocatalysts and supports for the ORR and the oxygen evolution reaction (OER, i.e. the anodic reaction of a proton exchange (or alkaline) membrane electrolyzer – PEME); (iii) hybrids systems, i.e. combination of man-made and biological systems, and cascades materials for various electrochemical systems.
M, N-doped carbonaceous electrocatalysts for the ORR, CO2RR and NRR
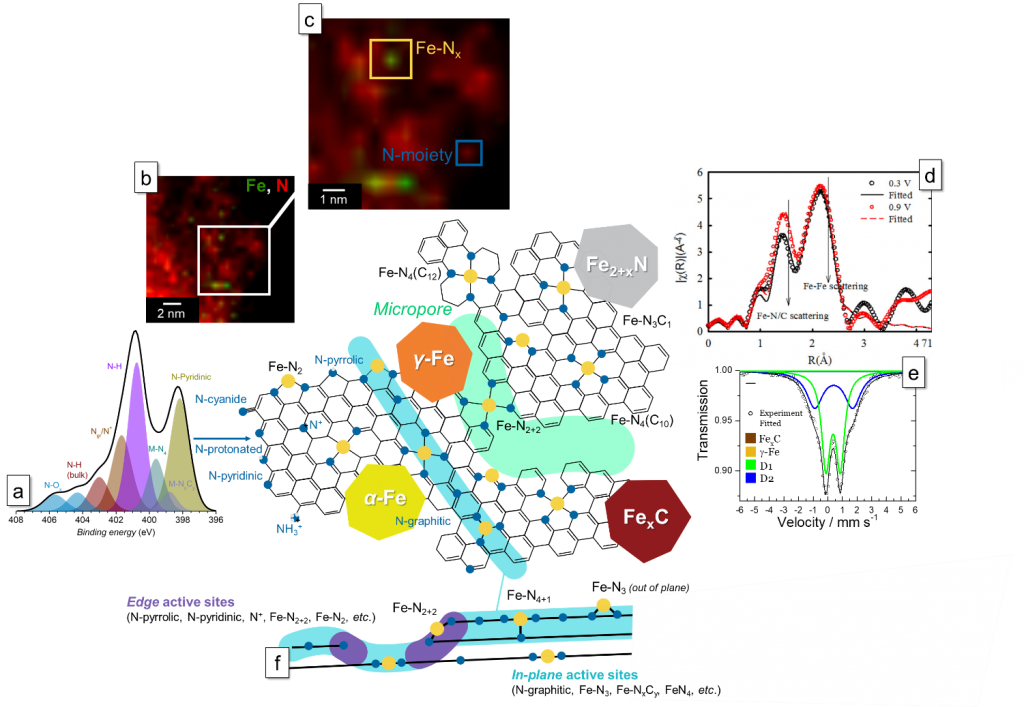
Figure 1. The Fe-N-C electrocatalyst and the different characterization methods used to assess its morphological & physico-chemical properties; (a) High resolution X-ray photoelectron spectroscopy in the N1s region of the electrocatalysts, obtained in collaboration with IRMI; (b – c) Electron energy loss spectrographs, obtained in collaboration with IMRI; (d) Fournier transform of an extended X-ray adsorption fine structure and (e) 57Fe Mossbauer pattern; (f) side view of the main graphitic plane presented in Figure 1, to evidence the difference between in-plane and edge active sites. Adapted from T. Asset, P. Atanassov, “Surface Chemistry, Morphology and Reactivity of Iron-Nitrogen-Carbon Catalysts for Proton Exchange Membrane Fuel Cells”, Joule, Just Accepted.
During the past decade, first in the university of New Mexico and, now, in the university of California Irvine, Atanassov’s group has been working on the next generation of nanomaterials for the PEMFC cathode, i.e. the non-PGM (platinum group metal) electrocatalysts. Those often consists of carbonaceous materials with atomically dispersed M (M = 3d transition metal) and N(itrogen), often referred as M-N-C, here synthesized by the sacrificial support method (SSM), which consist of the templating the material by silica, to be etched during the synthesis process using HF. The resulting material presents a wide variety of active sites (i.e. Fe-Nx, N-pyridinic, N-pyrrolic, etc.) along with a porous structure. Those materials mechanistic have been widely investigated by Atanassov’s group, i.e. reactivity of the different moieties, the role of the pH, the effect of the different pyrolysis, the involvement of the proton in the rate determining step, etc, as well as their activity and durability, in collaboration with numerous university such as the university of Grenoble and the university of Montpellier (France), the technical university of Berlin (Germany), the London imperial college (United Kingdom), etc.
Figure 2. Kinetic Isotope Effect (KIE) for Fe-N-C electrocatalysts. The KIE was determined by assessing the electrocatalysts reactivity in D2O and H2O based electrolyte (if the proton is involved in the rate determining step, replacing H+ by D+ should notably impact the kinetics). The absence of KIE at low overpotential indicate that the rate determining step is H+ independent in this potential region, whereas it is H+ dependent, likely from the side O2 → H2O2 reaction, at high overpotentials. Adapted from Y. Chen, T. Asset, R. Lee, K. Artyushkova, P. Atanassov, “Kinetic Isotopic Effect Studies of Iron-Nitrogen-Carbon Electrocatalysts for Oxygen Reduction Reaction”, J. Phys. Chem. C 123 (2019) 11476 – 11483 DOI: 10.1021/acs.jpcc.9b01480.
The investigation, and use, of such materials, recently extended to other mechanisms, e.g. the CO2 reduction reaction (CO2RR) and the nitrogen reduction reaction (NRR). Indeed, finding good electrocatalysts for said reactions is essential, as (i) although not the main contributor to the greenhouse effect, the CO2 concentration fluctuations (i.e. from 250 to 400 ppm) are greatly responsible for the global warming. Its reduction could lead to the formation of value added products (i.e. C2-alcohols, etc.) or syngas, for the Fischer-Tropsch and related energy generation processes; (ii) the NRR main product is ammonia (NH3) that plays a vital role as raw material in the manufacture of everyday life chemicals (fertilizers, pharmaceuticals, etc.); ammonia being currently mainly produced using the Haber-Bosch process (which operate at high pressure, high temperature and use H2 generated from fossil fuels). Both NRR and CO2RR can be achieved on M-N-C electrocatalysts or derived-structures (i.e. metal-free N-doped carbons, etc.) although the CO2RR is limited on such materials to the formation of CO, because of the low density of active sites. Hence, catalytic cascades (e.g. a combination of molecular, enzymatic and metallic/carbonaceous materials where each component catalyzes one step of the reaction) are also studied, to go beyond the CO formation, similarly to studies carried out in our group on glycerol oxidation by catalytic cascades. Other materials are under investigation, such as MXenes, Cu-based electrocatalyst and Mo, Fe-based electrocatalysts (in an attempt to mimic the nitrogenase Fe-Mo cofactor).
Another category of non-PGM materials that has been explored by Atanassov’s group are the Ni-based electrocatalysts for the hydrogen oxidation reaction (HOR, i.e. the most common anodic reaction of an alkaline (or proton exchange) membrane fuel cell) and hydrazine oxidation. Such study led to the conception of the first FC vehicle with both an anode and a cathode PGM free, in collaboration with Daihatsu.
Figure 3. Investigating the nature of the CO2RR actives sites on M-N-C electrocatalysts; (a – c) N1s high resolution x-ray photoelectron spectra of Fe, Co & Ni-N-C in ultra-high vacuum (UHV) and under CO2 atmosphere; (d) difference between XPS N1s spectra of the Fe, Co & Ni-N-C electrocatalysts in CO2 atmosphere and UHV, with the associated structures (an increase of the signal under the dark baseline means that the CO2 strongly adsorb on those specific sites). The light blue boxes indicate the binding region of different moieties; (e) Schematic representation of the different preferential adsorption sites for CO2 as a function of M (M = Fe, Co, Ni). Adapted from T. Asset, S. T. Garcia, S. Herrera, N. Andersen, Y. Chen, E. J. Petersen, I. Matanovic, K. Artyushkova, J. Lee, S. D. Minteer, S. Dai, X. Pan, K. Chavan, S. C. Barton, P. Atanassov, “Investigating the Nature of the Active Sites for the CO2 Reduction Reaction on Carbon-Based Electrocatalysts”, ACS Catal. 9 (2019) 7668 – 7678, DOI: 10.1021/acscatal.9b01513.
Pt-based electrocatalysts and supports for the oxygen reduction reaction and oxygen evolution reaction.
Figure 4. How to improve the activity of a Pt-based electrocatalyst: (i) Order-based approaches, i.e. controlling the interatomic distance and, thus, the electronic structure of platinum atom, through alloying and preferential orientation (REE = rare earth elements, TM = transition metals); (ii) i.e. controlling the local disorder (coordination number & local strain) to modify Pt electronic structure. Adapted from A. Ly, T. Asset, I. Zenyuk, P. Atanassov, “Implementing complex nanostructured Pt-based electrocatalysts in proton exchange membrane fuel cells”, in preparation.
Because of its ‘close-to-optimal’ binding energy toward the oxygen reduction reaction intermediates, platinum remain one of the most promising electrocatalyst as a PEMFC cathode (despite its price and scarcity) and, as such, is still commonly used as nanoparticles dispersed onto a carbon support. However, there is still several hurdles to overcome to achieve its commercialization:
-
Despite being the best element for the ORR, Pt specific activity, e. the activity reported to the Pt surface, can still be improved (see Figure 4). To achieve this objective, the Pt electronic structure can be modified by alloying or shaping of the nanocrystallites or increasing the density of structural defects to obtain actives sites with an ideal coordination number & atomic distance with their closest neighbors. However, although those ‘exotic’, improved electrocatalysts, exhibit tremendous performances for the ORR in liquid electrolytes, said performances do not often transpose to the proton exchange membrane fuel cell itself. Identifying the limitations of those electrocatalysts in solid electrolytes (e.g. PEMFCs) is one of Atanassov’s group objective.
Figure 5. Kinetic Isotope Effect (KIE) for carbon-supported Pt. The KIE was determined by assessing the electrocatalysts reactivity in D2O and H2O based electrolyte (if the proton is involved in the rate determining step, replacing H+ by D+ should notably impact the kinetics). The absence of KIE for high nanoparticles density on the carbon support indicates that, in such conditions, the rate determining step is H+ independent, whereas it is H+ dependent for a low nanoparticle density on the carbon surface. Adapted from M. Rezaei Talarposhti, T. Asset, S. T. Garcia, Y. Chen, S. Herrera, S. Dai, E. J. Peterson, K. Artyushkova, I. Zenyuk, P. Atanassov, “Kinetic Isotope Effect as a Tool to Investigate the Oxygen Reduction Re-action on Pt-based Electrocatalysts”, in preparation.
-
The activity is also dependent of the Pt dispersion on the support, hence requiring a better understanding of the support-electrocatalyst interactions. Said mechanisms are under investigation through KIE and near-ambient pressure X-ray photoelectron spectroscopy on Pt-poly and on carbon-supported Pt.
-
Another hurdle observed for Pt-based electrocatalysts are induced by the lack of stability of their support, e. carbon can thermodynamically oxidize in acidic environment at E > 0.2 V vs. RHE and its oxidation is catalyzed by Pt at E < 1.0 V vs. RHE. Modified carbons (e.g. fluorine-doped) or metallic oxides (TiO2, SnO2, etc.) are a viable alternative, because of their intrinsically higher stability. However, the synthesis of the latter has to be optimized to achieve a good compromise between stability and conductivity.
A viable alternative to TiO2, and a promising candidate as a support for the IrOx nanoparticles used as electrocatalysts for the oxygen evolution reaction (OER), is TiN. Indeed, this material exhibit a great electrochemical stability (because of the presence of a native TiO2/TiOxNy film on its surface) while maintaining a bulk electronic conductivity. It can also be shaped, via different synthesis processes, to insure a high specific surface.
Atanassov’s group presents a world-class expertise (i) in synthesis of non-PGM and PGM-group electrocatalysts for the oxygen reduction reaction, hydrogen oxidation reaction, etc. of carboneous and non-carbon based supports; (ii) in electrochemical methods, characterization, from liquid electrolyte to proton exchange and alkaline membrane fuel cells and (iii) in characterization techniques such as X-ray photoelectron spectroscopy, etc. In addition to the topic listed above, Atanassov’s group also has interest in (i) Cu-based electrocatalysts for the CO2RR, (ii) Al-air batteries and (iii) biofuel cells, artificial biofilms and hydrides systems, i.e. combination of man-made and biological systems (see Y. Liu and P. Atanassov, Charge Transfer at Biotic/Abiotic Interfaces in Biological Electrocatalysis, Current Opinion in Electrochemistry, just accepted).
Relevant publications:
-
Jaouen, F.; Herranz, J.; Lefevre, M.; Dodelet, J. P.; Kramm, U. I.; Herrmann, I.; Bogdanoff, P.; Maruyama, J.; Nagaoka, T.; Garsuch, A.; Dahn, J. R.; Olson, T.; Pylypenko, S.; Atanassov, P.; Ustinov, E. A. Cross-laboratory experimental study of non-noble-metal electrocatalysts for the oxygen reduction reaction. ACS Appl. Mater. Interfaces 2009, 1 (8), 1623–1639.
-
Jia, Q.; Ramaswamy, N.; Tylus, U.; Strickland, K.; Li, J.; Serov, A.; Artyushkova, K.; Atanassov, P.; Anibal, J.; Gumeci, C.; Barton, S. C.; Sougrati, M. T.; Jaouen, F.; Halevi, B.; Mukerjee, S. Spectroscopic insights into the nature of active sites in iron–nitrogen–carbon electrocatalysts for oxygen reduction in acid. Nano Energy 2016, 29, 65–82.
-
Chen, Y.; Matanovic, I.; Weiler, E.; Atanassov, P.; Artyushkova, K. Mechanism of oxygen reduction reaction on transition metal–nitrogen–carbon catalysts: establishing the role of nitrogen-containing active sites. ACS Appl. Energy Mater. 2018, 1 (11), 5948–5953.
-
Artyushkova, K.; Serov, A.; Rojas-Carbonell, S.; Atanassov, P. Chemistry of multitudinous active sites for oxygen reduction reaction in transition metal-nitrogen-carbon electrocatalysts. J. Phys. Chem. C 2015, 119 (46), 25917–25928.
-
Rojas-Carbonell, S.; Artyushkova, K.; Serov, A.; Santoro, C.; Matanovic, I.; Atanassov, P. Effect of pH on the activity of platinum group metal-free catalysts in oxygen reduction reaction. ACS Catal. 2018, 8 (4), 3041–3053.
-
Roy, A.; Talarposhti, M. R.; Normile, S. J.; Zenyuk, I. V.; De Andrade, V.; Artyushkova, K.; Serov, A.; Atanassov, P. Nickel-Copper Supported on a Carbon Black Hydrogen Oxidation Catalyst Integrated into an Anion-Exchange Membrane Fuel Cell. Sustain. Energy Fuels 2018, 2 (10), 2268–2275.
-
Asset, T.; Roy, A.; Sakamoto, T.; Padilla, M.; Matanovic, I.; Artyushkova, K.; Serov, A.; Maillard, F.; Chatenet, M.; Asazawa, K.; Tanaka, H.; Atanassov, P. Highly Active and Selective Nickel Molybdenum Catalysts for Direct Hydrazine Fuel Cell. Electrochim. Acta 2016, 215, 420–426.
-
Asset, T.; Job, N.; Busby, Y.; Crisci, A.; Martin, V.; Stergiopoulos, V.; Bonnaud, C.; Serov, A.; Atanassov, P.; Dubau, L.; Maillard, F. Porous Hollow PtNi/C Electrocatalysts: Carbon Support Considerations to Meet Performance and Stability Requirements. ACS Catal. 2018, 8, 893–903.
-
Andersen, N. I.; Artyushkova, K.; Matanović, I.; Hickey, D. P.; Minteer, S. D.; Atanassov, P. Spectro-Electrochemical Microfluidic Platform for Monitoring Multi-Step Cascade Reactions. ChemElectroChem 2019, 6 (1), 246–251.
-
Abdellaoui, S.; Seow Chavez, M.; Matanovic, I.; Stephens, A. R.; Atanassov, P.; Minteer, S. D. Hybrid Molecular/Enzymatic Catalytic Cascade for Complete Electro-Oxidation of Glycerol Using a Promiscuous NAD-Dependent Formate Dehydrogenase from: Candida Boidinii. Chem. Commun. 2017, 53 (39), 5368–5371.
-
Chen, Y; Asset, T; Lee, R; Artyushkova, K; Atanassov, P; “Kinetic Isotopic Effect Studies of Iron-Nitrogen-Carbon Electrocatalysts for Oxygen Reduction Reaction”, J. Phys. Chem. C 123 (2019) 11476 – 11483
-
Asset, T; Garcia, S. T; Herrera, S; Andersen, N; Chen, Y; Petersen, E. J; Matanovic, I; Artyushkova, K; Lee, J; Minteer, S. D; Dai, S; Pan, X; Chavan, K; Barton, S. C; Atanassov, P; “Investigating the Nature of the Active Sites for the CO2 Reduction Reaction on Carbon-Based Electrocatalysts”, ACS Catal. 9 (2019) 7668 – 7678