Autophagy Regulators and Melanogenesis
Melanin, a skin pigment that protects the skin and eyes from the harmful effects of UV, is synthesized in a specialized lysosome-related organelle called the melanosome. Melanosome maturation is precisely controlled by the sequential delivery of enzymes that synthesize melanin to the developing organelle such that melanin synthesis only occurs in the stage III or IV melanosome. Defects in melanosome maturation are linked to skin cancer susceptibility and skin pigment variation, highlighting the intrinsic biological relevance of this pathway. While extensive studies have identified vesicle transport pathways that deliver proteins to the melanosome, little is known about what directs transport vesicles to fuse with the developing melanosome. Recent work from our group has identified an unappreciated role for genes that regulate autophagy in the formation/maturation of the melanosome. The goal of this project is to determine is how do genes that regulate autophagy also regulate melanosome biogenesis? This work focuses on three key questions: 1) what determines whether these putative autophagy regulators control the formation of the autophagosome or melanosome? 2) how do these genes direct specific proteins to the melanosome? 3) what role do these genes play in coordinating melanosome formation with the transcription of genes that regulate melanogenesis? Completion of the proposed studies will not only enhance our fundamental understanding of how intracellular vesicle transport is coordinated and compartmentalized, but will also lead to the design of pharmacologic agents to stimulate melanosome maturation. These new drugs could then be used to induce melanin production in the skin of patients who are susceptible to UV induced skin cancer, reducing the disease burden of the most common type of cancer in the United States.
RhoJ/PAK1 Regulates Melanoma Invasion and Chemoresistance
Melanoma tumors metastasize at an early stage of development and are notoriously resistant to chemotherapeutics. We recently utilized a systems-level RNAi screening approach to uncover the molecular regulators of melanoma chemoresistance. These studies determined that RhoJ and its downstream kinase PAK1 suppress pathways in melanoma cells that sense DNA damage. Additional studies revealed that RhoJ and PAK1 also regulate melanoma invasion by modulating actin cytoskeletal dynamics. In this project, we seek to determine how RhoJ/PAK1 suppresses DNA damage sensing and determine whether Pak1 suppresses DNA damage sensing in vivo. Additional studies focus on identifying PAK kinase targets that modulate melanoma cell invasion and verify that PAK kinases regulate melanoma metastasis in vivo. We are also initiating translational studies to determine whether PAK kinases are selectively activated in poor prognosis/chemoresistant melanomas and investigate whether PAK inhibitors may be useful in the treatment of melanoma.
Define how ALDH1A1 Regulates Melanogenesis
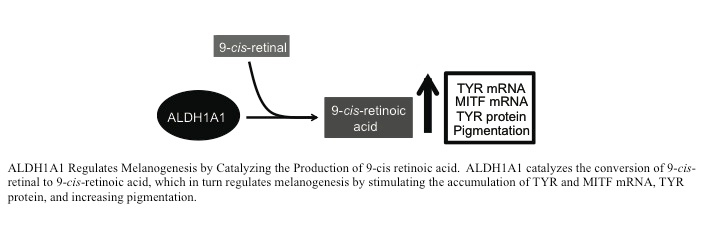
Aldehyde dehydrogenase 1A1 (ALDH1A1), an enzyme that catalyzes the conversion of retinal to retinoic acid, plays pleiotropic roles in melanogenesis, stem cell maintenance, and detoxification of UV-induced lipid aldehydes. Recent studies have utilized a combination of RNAi and pharmacologic approaches to identify specific ALDH1A1 substrates and products that regulate melanogenesis. Neither the ALDH1A1 product all-trans retinoic acid nor 4-hydroxy-2-nonenal, a UV-induced ALDH substrate, stimulated melanogenesis. In contrast, the ALDH1A1 substrate 9-cis retinal and its corresponding product 9-cis retinoic acid potently induced the accumulation of melanin and MITF and Tyrosinase mRNA. The addition of potent ALDH1A inhibitors, cyanamide and Angeli’s salt, suppressed Tyrosinase and MITF mRNA accumulation in vitro and also melanin accumulation in skin equivalents, suggesting that 9-cis retinoids regulate melanogenesis in the intact epidermis. Current studies are focused on how retinoids (all-trans retinoic acid) and rexinoids (9-cis retinoic acid) regulates melanogenesis and define the physiologic roles of 9-cis retinoic acid in melanogenesis. These studies will eventually lead to the rational design of pigment stimulatory agents that may have clinical utility in the treatment of hypopigmentary disorders, such as vitiligo.