“All Disease Begins in the Gut”
– Hippocrates
-
We are what we eat. Our goal is to understand how dietary nutrient processing by organs and gut microbiome contributes to metabolic disorders (obesity, diabetes) and liver disease (MASLD, MAT-ALD, liver cancer). We focus on human-relevant, common nutrients such as fructose, alcohol, fibers, and various fats and utilize analytical chemistry techniques including mass spectrometry-based metabolomics, lipidomics, and stable isotope tracing in animal models (mice, pigs) and human patients. Check out my podcast interview about fructose here: https://www.youtube.com/watch?v=vCD5Fct7ZzU
Members
Cholsoon Jang, PhD: Principal Investigator
Cholsoon received his PhD at Harvard Medical School. Then, he completed his postdoctoral fellow at Princeton University. He joined UCI in 2020 May as an Assistant Professor. He likes to bike, walk and watch animal videos.
Sunhee Jung, PhD: NRF & ADA Postdoctoral Fellow
Sunhee obtained her PhD in Chemistry at Sungkyunkwan University. She has expertise in analytical chemistry and nutrient metabolism. She obtained postdoctoral fellowships from the National Research Foundation (NRF) of Korea and the American Diabetes Association (ADA). She likes sunset and music and believes that sincerity is essential for every step in life.
Hosung Bae, PhD: NRF Postdoctoral Fellow
Hosung obtained his PhD in Biomedical Sciences and Engineering at KAIST in the laboratory of Dr. Gou Young Koh. He has expertise in bioinformatics and metabolic disease. He obtained a postdoctoral fellowship from the National Research Foundation (NRF) of Korea. He enjoys spending his free time with his kids, which he also regards it as science.
Won-Suk Song, PhD: KHIDI Postdoctoral Fellow
Won-Suk obtained his PhD in Chemical and Biological Engineering at Seoul National University in the laboratory of Prof. Byung-Gee Kim. He has expertise in biochemistry and host-microbe interaction. He obtained a postdoctoral fellowship from Korean Health Industry Development Institute (KHIDI). He likes watching soccer games and funny YouTube videos.
Wonsuk Choi, MD/PhD: Postdoctoral Fellow
Wonsuk obtained his PhD in Graduate School of Medical Science and Engineering at KAIST in the laboratory of Dr. Hail Kim. Then, he worked as a Clinical Assistant Professor in the Division of Endocrinology and Metabolism at Chonnam National University Hwasun Hospital. He has expertise in endocrinologic diseases with focus on metabolic liver and kidney diseases. In his free time, he enjoys sports with his kids, as well as chatting and drinking coffee with his wife
Johnny Le: MD/PhD student, recipient of NIDDK F31, CMCF Interdisciplinary Opportunity Award, and Behrens Research Excellence Award
Johnny is an MD/PhD student, studying skin metabolism. He has expertise in immunology with a focus on autoimmunity. He received the CMCF Interdisciplinary Opportunity Award. In his free time, he enjoys traveling and spending time with his family outdoors.
Cuauhtemoc (Temoc) Ramirez: PhD Student, recipient of NIH-IMSD Fellowship, T32 Cancer Training Grant
Temoc is a PhD student interested in studying signaling and metabolic pathways in the progression of diseases. When not in lab, he enjoys playing board games and sports with his friends and family.
Yasmine Henna Alam: PhD Student
Yasmine is a PhD student interested in understanding host-microbial interactions in the metabolism of drugs and diet-derived compounds. She enjoys spending time with her friends and family, being outdoors, and playing with puppies.
Ian James Tamburini: PhD Student
Ian is a PhD student interested in using ‘omics to understand how microbially derived metabolites impact systemic physiology. Ian comes from Boston, where he worked in industry studying microbiota in agricultural animals. In addition to science, he plays jazz saxophone and enjoys biking and exploring Orange County.
Miranda Emiko Kelly: PhD Student
Miranda is a PhD student interested in the identification and investigation of metabolically relevant microproteins. Outside of the lab, she enjoys knitting, playing board games with friends and hiking.
Miranda Lopez: PhD Student
Miranda aka “Randa” obtained her Biological Sciences BS at UC Irvine and Human Nutrition MS at Cal Poly, San Luis Obispo. She is interested in bridging the research gap between nutrient metabolism, immuno-endocrinology and human health. When not in the lab, she can be phenotyped as a gym rat, Yelp Elite and coffee connoisseur.
Izabelle Le: Lab Manager/Technician
Izabelle is a recent graduate from UCSD, where she earned her BS in Biochemistry. She is currently interested in and enjoys recombinant DNA microbiology and learning about cancer metabolism. When she’s not in the lab, Izabelle spends her time dancing, crocheting, watching movies, and coffee shop hopping. Although STEM and research is her main pursuit, she often dabbles in the artistic side of herself.
Alina Chao:Bio199 Undergraduate Research Student (UROP Fellow)
Alina is an undergraduate at UC Irvine majoring in Biological Sciences with an interest in nutrient metabolism. In her free time, she enjoys the outdoors by going on hikes and walking her dog.
Sang Hee Park: Bio199 Undergraduate Research Student (UROP Fellow)
Sang Hee is an international undergraduate at UC Irvine with a major in Biological Sciences. She is interested in diabetes and how metabolism works. In her free time, she enjoys working out and listening to music.
Taekyung Kang: Bio199 Undergraduate Research Student (UROP Fellow)
Taekyung is an undergraduate student at UCI majoring in Biological Sciences. His research interests include nutrient metabolism, protein pathway, and biotechnology. He enjoys watching Netflix and loves to travel around.
Varvara Rubtsova: Bio199 Undergraduate Research Student (UROP Fellow)
Varvara is an undergraduate student at UCI majoring in Biological Sciences. She is interested in nutrient metabolism and the gut microbiota, and how what we consume affects our health. In her free time, she enjoys playing tennis or volleyball and playing the guitar.
Nikki Joyce Khong: Bio199 Undergraduate Research Student (UROP Fellow)
Nikki is an undergraduate student at UCI majoring in Biological Sciences. Her research interests include nutrient metabolism and NAFLD. In her free time, she enjoys watching new shows and learning how to cook.
Marie Kim: Bio199 Undergraduate Research Student
Marie is an undergraduate student studying Biological Sciences. She is interested in learning more about metabolic disease. She enjoys walking her dogs and going to the beach.
Jongwon Baek: Bio199 Undergraduate Research Student
Jongwon is an undergraduate student majoring in Biomedical Engineering: pre-med. He is interested in nutrient metabolism, especially the intricate mechanisms connecting dietary choices with various health conditions. He enjoys playing soccer and playing video games during his free time.
Elena Marie Moyer: Bio199 Undergraduate Research Student
Elena is an undergraduate student at UCI majoring in Biological Sciences. She is interested in nutrient metabolism as well as the gut microbiome and their correlation to disease. In her free time, she enjoys going to cafes and museums.
Ariel Tsai: Bio199 Undergraduate Research Student (UROP Fellow)
Ariel is an undergraduate student at UCI studying Biological Sciences. She is interested in nutrient metabolism, specifically the aspects of nutritional compounds in foods we consume that affect metabolic diseases. She likes baking and cooking in her free time
Daniel Onofre: Bio199 Undergraduate Research Student
Daniel is an undergraduate at UC Irvine majoring in Biological Sciences with an interest in gut microbiome health. In his free time, he loves to train Brazilian jiu-jitsu and go to the gym.
Yeojin Kim: Bio199 Undergraduate Research Student (UROP Fellow)
Yeojin is an undergraduate student at UCI majoring in Biological Sciences. She is interested in the interconnection between nutrient metabolism and various health concerns. In her free time, she enjoys traveling and discovering new restaurants and aesthetic cafes.
Nicholas D’Sa: Bio199 Undergraduate Research Student
Nicholas is an undergraduate student at UCI majoring in Public Health Sciences. He is interested in uncovering the mechanisms of metabolic disorders to develop novel therapies and diagnostic methods. In his free time, he enjoys playing piano, guitar, and tennis.
Former members
- Shea Skenderian (Former: Summer research intern 2020-2021, Current: Undergraduate at UC Berkeley)
- Lavina Mathur (Former: lab manager/technician 2020-2021, Current: Lab manager at Stanford U)
- Lily Tong (Former: Bio199 student 2021-2022, Current: Undergraduate at UC Irvine)
- Grace Park (Former: Post-BA researcher 2020-2022, Current: PhD graduate student at U Penn)
- Kamile Marie Macandili (Former: Bio199 student 2021-2022)
- Hayoung Choi (Former: Bio199 student 2022-2023, Current: Yonsei University)
- Lauren Hoffner (Former: lab manager/technician 2021-2023, Current: PhD graduate student at Scripps)
- Raymond Kim (Former: Post-BA researcher 2020-2023, Current: Medical Student at University of Nevada)
- Sang-Guk Lee (Former: Visiting Scholar 2022-2024, Current: Associate Professor in Yonsei University College of Medicine)
- Jessie Kim (Former: Bio199 Post Baccalaureate Research Student 2023-2024, Current: Pre-med)
Research
1. Nutrients & Metabolic disorders and cancers
There are numerous associations between foods and diseases (e.g., the link between soda drinking and fatty liver). In most cases, however, the causality is elusive or underlying mechanisms are controversial. This problem likely originates from our incomplete understanding of how nutrients are processed in our body.
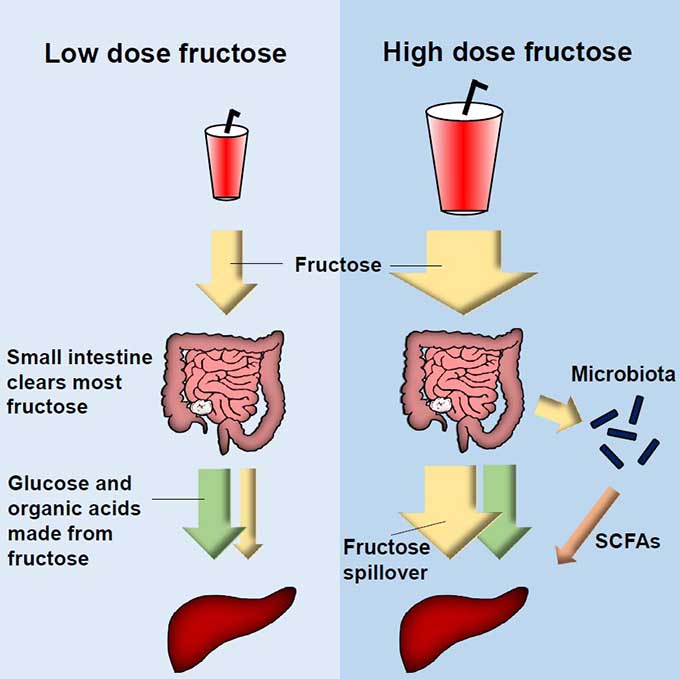
One example is the metabolism of dietary fructose, a risk factor for obesity, diabetes, and fatty liver. Based on gene expression, it was believed that the liver is the sole site of fructose breakdown. However, our recent works showed that the small intestine clears most physiological doses of fructose even before fructose reaches the liver. Only high fructose doses overwhelm the intestine’s capacity, resulting in fructose spillover to the liver and also to the colonic gut microbiome. Such spillover causes excessive fatty acid synthesis in the liver and gut dysbiosis. Now, we study how fructose and other nutrients such as dietary fibers or alcohol interacts with and worsens/prevents systemic disorders (diabetes, obesity, fatty liver) and liver cancers.
Jang C et al. Cell Metabolism (2018), Zhao S*, Jang C* et al. Nature (2020), Jang C*, Wada S* et al. Nature Metabolism (2020).
2. Inter-organ Metabolic Communications
In our body, no single organ is isolated. Organs share metabolites via circulation dynamically to achieve whole-body balance and communications. One famous example is the Cori cycle where muscle burns glucose and makes lactate, which is converted back into glucose by the liver to feed skeletal muscle.
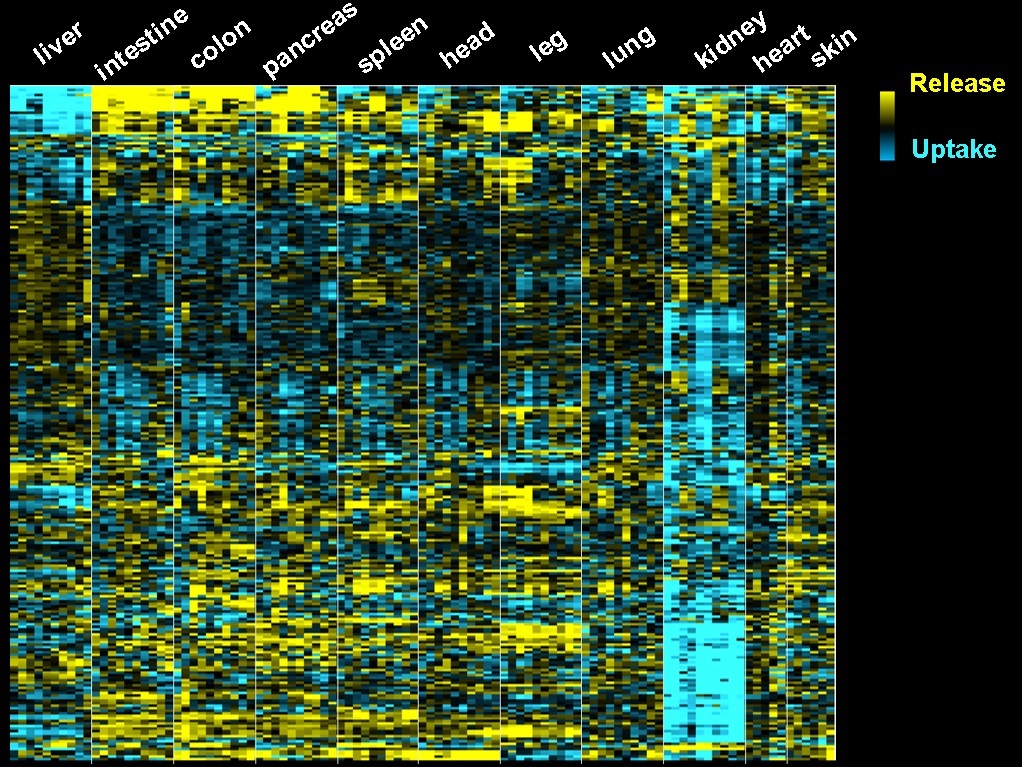
To understand this process comprehensively, we measured metabolite concentrations in arterial blood (organ input) and venous blood (organ output) to quantitate organ-specific metabolite release and uptake fluxes. For this method, we use large animals such as pigs or humans. We recently also applied this method even in mice to understand brown fat, muscle and kidney metabolism.
Using this method, we generated datasets containing over 700 significant cases of organ-specific metabolite production or consumption in pigs. Now, we apply this technology in various disease animal models to understand how inter-organ metabolic crosstalk is maintained in health and goes awry to cause disease.
Jang C et al. Cell Metabolism (2019), Murashige D*, Jang C* et al. Science (2020), Li et al. Nature Metabolism (2022), Park et al. Nature Metabolism (2023).
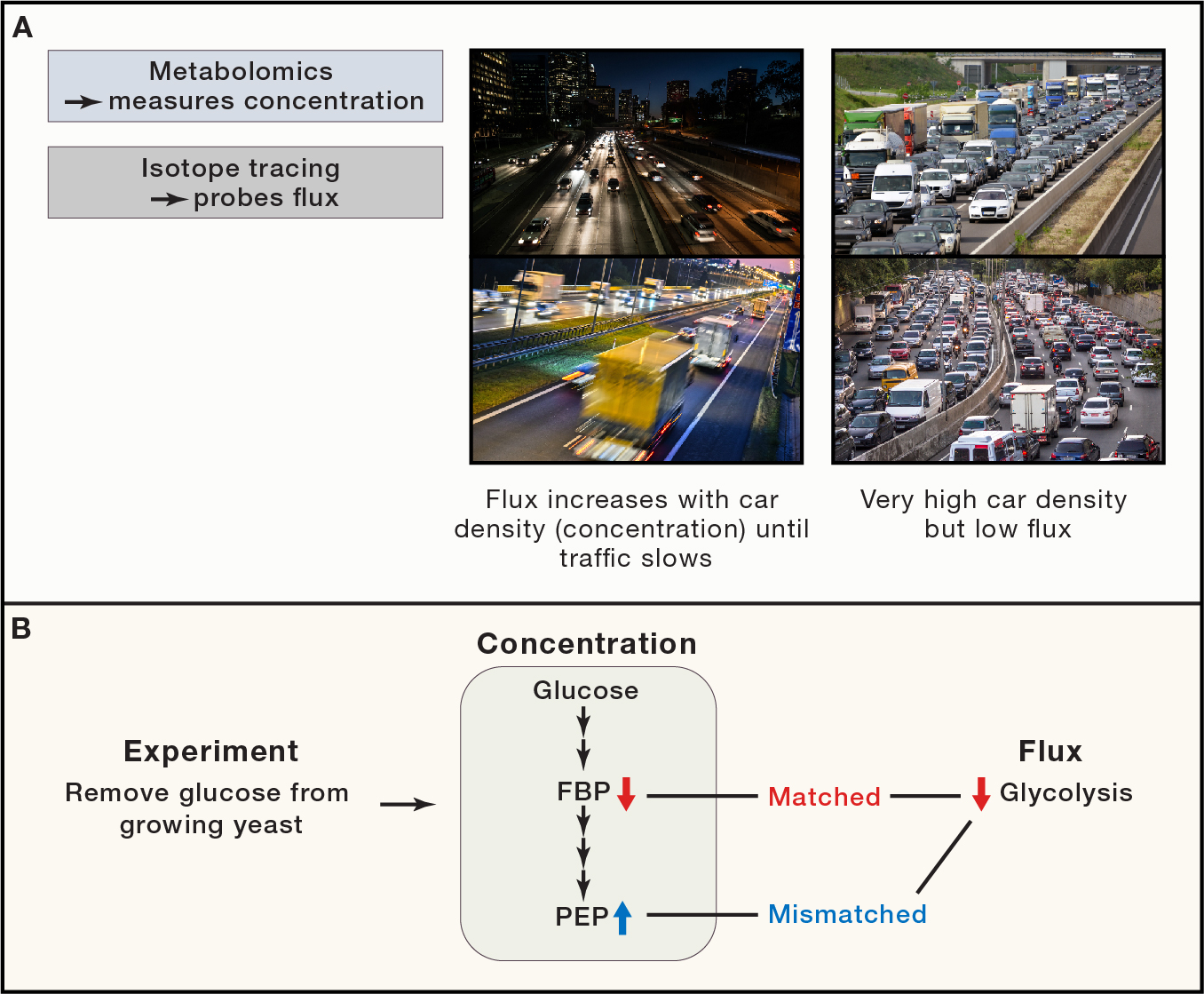
3. Metabolomics and Isotope Tracing Technology Development
Our technological platform is metabolomics, non-radioactive stable isotope tracing and bioinformatic analysis. We have in-house high-sensitivity, high-resolution Orbitrap LC-MS instruments. By developing and optimizing new measurement methods, we aim to identify bioactive metabolites and quantitate biologically important metabolic pathway activities.
Jang C et al. Cell (2018). Zeng X et al. Cell (2022).
Selected Publications (Jang lab underlined)
2023
Doshi MB, Lee N, Tseyang T, Ponomarova O, Goel HL, Spears M, Li R, Zhu LJ, Ashwood C, Simin K, Jang C, Mercurio AM, Walhout AJM, Spinelli JB, Kim D. (2023). Disruption of sugar nucleotide clearance is a therapeutic vulnerability of cancer cells. Nature. 623(7987):625-632.
Green CL, Trautman ME, Chaiyakul K, Jain R, Alam YH, Babygirija R, Pak HH, Sonsalla MM, Calubag MF, Yeh CY, Bleicher A, Novak G, Liu TT, Newman S, Ricke WA, Matkowskyj KA, Ong IM, Jang C, Simcox J, Lamming DW. (2023). Dietary restriction of isoleucine increases healthspan and lifespan of genetically heterogeneous mice.
Cell Metabolism. 35(11):1976-1995.
Cho S, Chun Y, He L, Ramirez CB, Ganesh KS, Jeong K, Song J, Cheong JG, Li Z, Choi J, Kim J, Koundouros N, Ding F, Dephoure N, Jang C, Blenis J, Lee G. (2023). FAM120A couples SREBP-dependent transcription and splicing of lipogenesis enzymes downstream of mTORC1. Molecular Cell. 83(16):3010-3026.
Park G*, Haley JA*, Le J*, Jung SM*, Fitzgibbons TP, Korobkina ED, Li H, Fluharty SM, Chen Q, Spinelli JB, Trivedi CM, Jang C#, Guertin DA#. (2023). Quantitative analysis of metabolic fluxes in brown fat and skeletal muscle during thermogenesis. Nature Metabolism. 5(7): 1204-1220.
Zhu X, Wang Y, Soaita I, Lee HW, Bae H, Boutagy N, Bostwick A, Zhang RM, Bowman C, Xu Y, Trefely S, Chen Y, Qin L, Sessa W, Tellides G, Jang C, Snyder NW, Yu L, Arany Z, Simons M. (2023). Acetate controls endothelial-to-mesenchymal transition. Cell Metabolism. S1550-4131(23)00203-6.
Lin L, Jung KM, Lee HL, Le J, Colleluori G, Wood C, Palese F, Squire E, Ramirez J, Su S, Torrens A, Fotio Y, Tang L, Yu C, Yang Q, Huang L, DiPatrizio N, Jang C, Cinti S, Piomelli D. (2023). Adolescent exposure to low-dose THC disrupts energy balance and adipose organ homeostasis in adulthood. Cell Metabolism. S1550-4131(23)00179-1.
Smith JG, Koronowski KB, Mortimer T, Sato T, Greco CM, Petrus P, Verlande A, Chen S, Samad M, Deyneka E, Mathur L, Blazev R, Molendijk J, Kumar A, Deryagin O, Vaca-Dempere M, Sica V, Liu P, Orlando V, Parker BL, Baldi P, Welz PS, Jang C, Masri S, Benitah SA, Muñoz-Cánoves P, Sassone-Corsi P. (2023). Liver and muscle circadian clocks cooperate to support glucose tolerance in mice. Cell Reports. 42(6):112588.
Dragan M, Chen Z, Li Y, Le J, Sun P, Haensel D, Sureshchandra S, Pham A, Lu E, Pham KT, Verlande A, Vu R, Gutierrez G, Li W, Jang C, Masri S, Dai X. (2023). Ovol1/2 loss-induced epidermal defects elicit skin immune activation and alter global metabolism. EMBO Reports. e56214.
Blair MC, Neinast MD, Jang C, Chu Q, Jung JW, Axsom J, Bornstein MR, Thorsheim C, Li K, Hoshino A, Yang S, Roth Flach RJ, Zhang BB, Rabinowitz JD, Arany Z. (2023). Branched-chain amino acid catabolism in muscle affects systemic BCAA levels but not insulin resistance. Nature Metabolism. 5(4):589-606.
Guan D, Bae H, Zhou D, Chen Y, Jiang C, La CM, Xiao Y, Zhu K, Hu W, Trinh TM, Liu P, Xiong Y, Cai B, Jang C, Lazar MA. (2023). Hepatocyte SREBP signaling mediates clock communication within the liver. J Clin Invest. 133(8):e163018.
Park S, Belfoul AM, Rastelli M, Jang A, Monnoye M, Bae H, Kamitakahara A, Giavalisco P, Sun S, Barelle PY, Plows J, Jang C, Fodor A, Goran MI, Bouret SG. (2023). Maternal low-calorie sweetener consumption rewires hypothalamic melanocortin circuits via a gut microbial co-metabolite pathway. JCI Insight. 8(10):e156397.
2022
Le J, Jang C. (2022). Genetic variation in sugar metabolism confers a protective metabolic profile. Gastroenterology S0016-5085(22)00014-2.
Li X, Hui S, Mirek ET, Jonsson WO, Anthony TG, Lee WD, Zeng X, Jang C#, Rabinowitz JD#. (2022). Circulating metabolite homeostasis achieved through mass action. Nature Metabolism 1:141-152.
Green CL, Pak HH, Richardson NE, Flores V, Yu D, Tomasiewicz JL, Dumas SN, Kredell K, Fan JW, Kirsh C, Chaiyakul K, Murphy ME, Babygirija R, Barrett-Wilt GA, Rabinowitz J, Ong IM, Jang C, Simcox J, Lamming DW. (2022). Sex and genetic background define the metabolic, physiologic, and molecular response to protein restriction. Cell Metabolism 34(2):209-226.
Alam YH, Kim R, Jang C. (2022). Metabolism and Health Impacts of Dietary Sugars. J Lipid Atheroscler. 11(1):20-38.
Jung SM*, Le J*, Doxsey WG*, Haley JA*, Park G, Guertin DA#, Jang C#. (2022). Stable Isotope Tracing and Metabolomics to Study In Vivo Brown Adipose Tissue Metabolic Fluxes. Methods Mol Biol. 2448:119-130.
Gosis BS, Wada S, Thorsheim C, Li K, Jung S, Rhoades JH, Yang Y, Brandimarto J, Li L, Uehara K, Jang C, Lanza M, Sanford NB, Bornstein MR, Jeong S, Titchenell PM, Biddinger SB, Arany Z. (2022). Inhibition of nonalcoholic fatty liver disease in mice by selective inhibition of mTORC1. Science 376(6590):eabf8271.
Wu Y, Wong CW, Chiles EN, Mellinger AL, Bae H, Jung S, Peterson T, Wang J, Negrete M, Huang Q, Wang L, Jang C, Muddiman DC, Su X, Williamson I, Shen X. (2022). Glycerate from intestinal fructose metabolism induces islet cell damage and glucose intolerance. Cell Metabolism S1550-4131(22)00189-9.
Nguyen HP, Villivalam SD, Jung BC, You D, Lin F, Yi D, Pi A, Ma K, Jung S, Park SH, Jang C, Sul HS, Kang S. (2022). AIFM2 is Required for High-Intensity Aerobic Exercise by Promoting Glucose Utilization. Diabetes doi: 10.2337/db21-1114.
Petrus P*, Cervantes M*, Samad M, Sato T, Chao A, Sato S, Koronowski KB, Park G, Alam Y, Mejhert N, Seldin MM, Monroy Kuhn JM, Dyar KA, Lutter D, Baldi P, Kaiser P, Jang C, Sassone-Corsi P. (2022). Tryptophan metabolism is a physiological integrator regulating circadian rhythms. Molecular Metabolism 29;64:101556.
Durairaj G, Demir Ö, Lim B, Baronio R, Tifrea D, Hall LV, DeForest JC, Lauinger L, Jebril Fallatah MM, Yu C, Bae H, Lin DW, Kim JK, Salehi F, Jang C, Qiao F, Lathrop RH, Huang L, Edwards R, Rychnovsky S, Amaro RE, Kaiser P. (2022). Discovery of compounds that reactivate p53 mutants in vitro and in vivo. Cell Chemical Biology S2451-9456(22)00275-6.
Jung S*, Bae H*, Song WS*, Jang C. (2022). Dietary Fructose and Fructose-Induced Pathologies. Annual Review of Nutrition 22;42:45-66.
Zeng X, Xing X, Gupta M, Keber FC, Lopez JG, Lee YJ, Roichman A, Wang L, Neinast MD, Donia MS, Wühr M#, Jang C#, Rabinowitz JD#. (2022). Gut bacterial nutrient preferences quantified in vivo. Cell 185:3441-3456.
Bae H, Lam K, Jang C. (2022). Metabolic flux between organs measured by arteriovenous metabolite gradients. Experimental & Molecular Medicine. 54:1354-1366.
Hokenson RE, Alam YH, Short AK, Jung S, Jang C, Baram TZ. (2022). Sex-dependent effects of multiple acute concurrent stresses on memory: a role for hippocampal estrogens. Front Behav Neurosci. 8;16:984494.
Murashige D, Jung JW, Neinast MD, Levin MG, Chu Q, Lambert JP, Garbincius JF, Kim B, Hoshino A, Marti-Pamies I, McDaid KS, Shewale SV, Flam E, Yang S, Roberts E, Li L, Morley MP, Bedi KC Jr, Hyman MC, Frankel DS, Margulies KB, Assoian RK, Elrod JW, Jang C, Rabinowitz JD, Arany Z. (2022). Extra-cardiac BCAA catabolism lowers blood pressure and protects from heart failure. Cell Metabolism S1550-4131(22)00398-9.
Flam E, Jang C, Murashige D, Yang Y, Morley MP, Jung S, Kantner DS, Pepper H, Bedi KC Jr, Brandimarto J, Prosser BL, Cappola T, Snyder NW, Rabinowitz JD, Margulies KB, Arany Z. (2022). Integrated landscape of cardiac metabolism in end-stage human nonischemic dilated cardiomyopathy. Nature Cardiovascular Research. 1(9):817-829.
2021
Cho S*, Lee G*, Pickering BF*, Jang C, Park JH, He L, Mathur L, Kim SS, Jung S, Tang HW, Monette S, Rabinowitz JD, Perrimon N, Jaffrey SR, Blenis J. (2021). mTORC1 promotes cell growth via m6A-dependent mRNA degradation. Molecular Cell 81:2064-2075.
Yu D*, Richardson NE*, Green CL, Spicer AB, Murphy ME, Flores V, Jang C, Kasza I, Nikodemova M, Wakai MH, Tomasiewicz JL, Yang SE, Miller BR, Pak HH, Brinkman JA, Rojas JM, Quinn WJ, Cheng EP, Konon EN, Haider LR, Finke M, Sonsalla M, Alexander CM, Rabinowitz JD, Baur JA, Malecki KC and Lamming DW. (2021). The adverse metabolic effects of branched-chain amino acids are mediated by isoleucine and valine. Cell Metabolism 33:905-922.
Park G, Jung SH, Wellen KE, Jang C. (2021). The interaction between the gut microbiota and dietary carbohydrates in nonalcoholic fatty liver disease. Experimental & Molecular Medicine 53:809-832.
Verlande A, Chun SK, Goodson MO, Fortin BM, Bae H, Jang C, Masri S. (2021). Glucagon regulates the stability of REV-ERBα to modulate hepatic glucose production in a model of lung cancer–associated cachexia. Science Advances 7(26):eabf3885.
Saiman Y, Shen TD, Lund PJ, Gershuni VM, Jang C, Patel S, Jung S, Furth EE, Friedman ES, Chau L, Garcia BA, Wu GD. (2021). Global microbiota-dependent histone acetylation patterns are irreversible and independent of short chain fatty acids. Hepatology 74:3427-3440.
Jung SM, Doxsey WG, Le J, Haley JA, Mazuecos L, Luciano AK, Li H, Jang C#, Gertin DA#. (2021). In vivo isotope tracing reveals the versatility of glucose as a brown adipose tissue substrate. Cell Reports 36(4):109459.
Koronowski KB#, Greco CM#, Huang H, Kim J, Fribourgh JL, Crosby P, Mathur L, Ren X, Partch CL, Jang C, Qiao F, Zhao Y, Sassone-Corsi P. Ketogenesis impact on liver metabolism revealed by proteomics of lysine β-hydroxybutyrylation. (2021). Cell Reports 36(5):109487.
Mathur L*, Jung S*, Jang C#, Lee G#. (2021). Quantitative analysis of m6A RNA modification by LC-MS. STAR Protocols 3:100724.
Basehore S, Bohlman S, Weber C, Swaminathan S, Zhang Y, Jang C, Arany Z, Clyne AM. (2021). Laminar flow on endothelial cells suppresses eNOS O-GlcNAcylation to promote eNOS activity. Circulation Research 129:1054-1066.
2020
Zhao S*, Jang C*, Liu L, Uehara K, Gilbert M, et al. (2020). Dietary fructose feeds hepatic lipogenesis via microbiota-derived acetate. Nature 579:586-591.
Jang C*,#, Wada S*, Yang S, Gosis B, Zeng X, et al. (2020). The small intestine shields the liver from fructose-induced steatosis. Nature Metabolism 2:586-593. #corresponding author
Bae H, Hong KY, Lee CK, Jang C, Lee SJ, et al. (2020). Angiopoietin-2-integrin α5β1 signaling enhances vascular fatty acid transport and prevents ectopic lipid-induced insulin resistance. Nature Communications 12;11(1):2980.
Yang L, Garcia Canaveras JC, Chen Z, Wang L, Liang L, Jang C, et al. (2020). Serine catabolism feeds NADH when respiration is impaired. Cell Metabolism 31:809-821.
Guan D, Xiong Y, Trinh TM, Xiao Y, Hu W, Jiang C, Dierichx P, Jang C, Rabinowitz JD, Lazar M. (2020). The hepatocyte clock and feeding control chronophysiology of multiple liver cell types. Science 369:1388-1394.
Hui S*,#, Cowan AJ*, Zeng X, Yang L, TeSlaa T, Li X, Bartman C, Zhang Z, Jang C, Wang L, Lu W, Rojas J, Baur J, Rabinowitz JD#. (2020). Quantitative fluxomics of circulating metabolites. Cell Metabolism 32:676-688.
Murashige D*, Jang C*, Neinast M, Edwards JJ, Cowan A, Hyman MC, Rabinowitz JD, Frankel DS, Arany Z. (2020). Comprehensive quantification of fuel use by the failing and nonfailing human heart. Science 370:364-368.
Skenderian S, Park G, Jang C. (2020). Organismal fructose metabolism in health and non-alcoholic fatty liver disease. Biology (Basel) 9(11):405.
Before 2020
Jang C, Hui S, Zeng X, Cowan AJ, Wang L, et al. (2019). Metabolite exchange between mammalian organs quantified in pigs. Cell Metabolism 30:596-606.
Lee CK, Jeong SH, Jang C, Bae H, Kim YH, et al. (2019) Tumor metastasis to lymph nodes requires YAP-dependent metabolic adaptation. Science 363:644-649.
Neinast M*, Jang C*, Hui S, Murashige DS, Chu Q, et al. (2019). Quantitative analysis of the whole-body metabolic fate of branched-chain amino acids. Cell Metabolism 29:417-429.
Kim B, Jang C, Dharaneeswaran H, Li J, Bhide M, et al. (2018) Endothelial pyruvate kinase M2 maintains vascular integrity. J. Clin. Invest. 128:4543-4556.
Jang C, Li C, Rabinowitz JD. (2018) Metabolomics and isotope tracing. Cell 173:822-837. (Review)
Jang C, Hui S, Lu W, Cowan AJ, Morscher RJ, et al. (2018) The small intestine converts dietary fructose into glucose and organic acids. Cell Metabolism 27:351-361.
Mirtschink P, Jang C, Arany Z, Krek W. (2018) Fructose metabolism, cardiometabolic risk, and the epidemic of coronary artery disease. Eur. Heart J. 39:2497-2505. (Review)
Guan D, Xiong Y, Borck PC, Jang C, Doulias PT, et al. (2018) Diet-Induced circadian enhancer remodeling synchronizes opposing hepatic lipid metabolic processes. Cell 174:831-842.
Lanaspa MA, Andres-Hernando A, Orlicky DJ, Cicerchi C, Jang C, et al. (2018) Ketohexokinase C blockade ameliorates fructose-induced metabolic dysfunction in fructose-sensitive mice. J. Clin. Invest. 128:2226-2238.
Hui S, Ghergurovich JM, Morscher RJ, Jang C, Teng X, et al. (2017) Glucose feeds the TCA cycle via circulating lactate. Nature 551:115-118.
Lee G, Zheng Y*, Cho S*, Jang C, England C, et al. (2017) Post-transcriptional regulation of de novo lipogenesis by mTORC1-S6K1-SRPK2 signaling. Cell 171:1545-1558.
Kim B*, Li J*, Jang C, Arany Z. (2017) Glutamine fuels proliferation but not migration of endothelial cells. EMBO J. 36:2321-2333.
Jang C*, Oh SF*, Wada S, Rowe GC, Liu L, et al. (2016) A branched chain amino acid metabolite drives vascular fatty acid transport and insulin resistance. Nature Medicine 22:421-426.
Wada S, Neinast M*, Jang C*, Ibrahim YH, Lee G, et al. (2016) The tumor suppressor FLCN mediates an alternate mTOR pathway to regulate browning of adipose tissue. Genes Dev. 30:2551-2564.
Rowe GC*, Raghuram S*, Jang C, Nagy JA, Patten IS, et al. (2014) PGC-1α induces SPP1 to activate macrophages and orchestrate functional angiogenesis in skeletal muscle. Circulation Res. 115:504-517.
Jang C and Arany Z. (2013) Metabolism: Sweet enticements to move. Nature 500:409-411. (News & views).
Patten IS*, Rana S*, Shahul S, Rowe GC, Jang C, et al. (2012) Cardiac angiogenic imbalance leads to peri-partum cardiomyopathy. Nature 485:333-338.
Kataru RP*, Kim H*, Jang C, Choi DK, Koh BI, et al. (2011) T lymphocytes negatively regulate lymph node lymphatic vessel formation. Immunity 34:96-107.
Jang C, Koh YJ, Lim NK, Kang HJ, Kim DH, et al. (2009) Angiopoietin-2 exocytosis is stimulated by sphingosine-1-phosphate in human blood and lymphatic endothelial cells. Arterioscler. Thromb. Vasc. Biol. 29:401-407.
Jang C, Lee G, Chung J. (2008) LKB1 induces apical trafficking of Silnoon, a monocarboxylate transporter, in Drosophila. J. Cell Biol. 183:11-17.
Jeon BH, Jang C, Han J, Kataru RP, Piao L, et al. (2008) Profound but dysfunctional lymphangiogenesis via vascular endothelial growth factor ligands from CD11b+ macrophages in advanced ovarian cancer. Cancer Res. 68:1100-1109.
Complete list of publications
- Pubmed: https://www.ncbi.nlm.nih.gov/pubmed/?term=cholsoon+jang
- Google Scholar: https://scholar.google.com/citations?user=Wsdt99kAAAAJ&hl=en&oi=ao
Lab News
- 5/1/2020. The Jang lab was born healthy, despite the COVID-19 pandemic.
- 5/1/2020. Cholsoon received Center for Epigenetic and Metabolism Pilot Award! Fall 2019 Newsletter (uci.edu)
- 5/1/2020. Cholsoon received UCI Microbiome Initiative Pilot Award! 2020 Pilot Project Awards – UCI Microbiome Initiative
- 6/22/2020. Cholsoon and Shogo’s paper is out in Nature Metabolism! https://www.nature.com/articles/s42255-020-0222-9
- 7/17/2020. Grace joined the lab.
- 7/27/2020. Lavina joined the lab.
- 7/28/2020. Cholsoon received AASLD Pinnacle Research Award! http://www.aasldfoundation.org/2020-research-and-career-development-award-recipients
- 8/7/2020. Q Exactive LC-MS started collecting the lab’s first data.
- 9/1/2020. Sunhee joined the lab.
- 10/1/2020. Johnny joined the lab.
- 10/1/2020. Hosung joined the lab.
- 10/1/2020. Cholsoon received Edward Mallinckrodt, Jr. Foundation Award! http://emallinckrodtfoundation.org/Current_Awardees.html
- 10/15/2020. Danielle and Cholsoon‘s paper is out in Science! https://science.sciencemag.org/content/370/6514/364
- 11/17/2020. Shea and Grace‘s paper is out in Biology! https://www.mdpi.com/2079-7737/9/11/405/htm
- 12/1/2020. Johnny and Remi in Xing Dai’s lab received CMCF Interdisciplinary Opportunity Award!
- 12/7/2020. Cholsoon received Southern California Research Center for ALPD and Cirrhosis Pilot Award! https://keck.usc.edu/alpd-and-cirrhosis-research-center/about/pilot-projects
- 3/29/2021. Cholsoon and Dr. Gina Lee’s project received Center for Cancer Systems Biology Pilot Award! https://ccbs.uci.edu/research/casb/pilot-projects/
- 3/29/2021. Temoc joined the lab.
- 4/21/2021. Cholsoon received American Cancer Society Institutional Research Grant! https://cancerresearch.uci.edu/acs-seed-grant-awardees/
- 4/30/2121. Both Sunhee and Hosung received 3-year postdoctoral fellowships!
- 5/13/2021. Cholsoon, with Drs. Yama Akbari and Robert Wilson, received Susan Samueli Integrative Health Institute (SSIHI) Pilot Studies Award! https://ssihi.uci.edu/education-research/pilot-studies-program/
- 5/20/2021. Grace and Sunhee‘s paper is out in Experimental and Molecular Medicine! https://www.nature.com/articles/s12276-021-00614-x
- 6/4/2021. Cholsoon, with Drs. Kaiser and Rychnovsky, received CFCCC Anti-Cancer Challenge Pilot Award! http://www.cancer.uci.edu/research/anti-cancer-challenge-research/anti-cancer-challenge-rfp
- 6/11/2021. Lavina left the lab for her new job as a Senior Research Associate at Prellis Biologics. Best luck!
- 6/14/2021. Yasmine joined the lab.
- 6/28/2021. Lauren joined the lab.
- 7/21/2021. We received our first R01 grant!
- 7/27/2121. The first research paper from the lab is published in Cell Reports! https://www.sciencedirect.com/science/article/pii/S2211124721008822
- 8/6/2021. Lavina and Sunhee‘s paper is out in STAR Protocol! https://www.sciencedirect.com/science/article/pii/S2666166721004317
- 9/23/2021. Temoc received the NIH-IMSD fellowship!
- 1/8/2022. Johnny’s Editorial is out in Gastroenterology! https://www.sciencedirect.com/science/article/pii/S0016508522000142?via%3Dihub
- 1/17/2022. Yasmine and Raymond‘s paper is out in J Lipid Athreoscler! https://pubmed.ncbi.nlm.nih.gov/35118020/
- 1/21/2022. A paper Cholsoon participated as a co-corresponding author is published in Nature Metabolism! https://www.nature.com/articles/s42255-021-00517-1
- 2/15/2022. Johnny‘s paper about stable isotope tracing in BAT is out in Methods Mol Biol! https://pubmed.ncbi.nlm.nih.gov/35167094/
- 3/6/2022. Cholsoon was invited to give a talk at the prestigious Forbeck Forum! Diet and Metabolic Therapeutics in Cancer – Forbeck Forums
- 6/8/2022. Johnny passed his advancement exam! Officially the first MD, PhD candidate in the lab!
- 8/15/2022. Temoc received T32 Cancer Research Training Award!
- 8/23/2022. Sunhee, Hosung, and Won-Suk‘s fructose review paper is published in the Annual Review of Nutrition! Dietary Fructose and Fructose-Induced Pathologies | Annual Review of Nutrition (annualreviews.org)
- 10/18/2022. Yasmine received TWO poster awards (one from judges and another from the audience) at UCI School of Medicine Grad Day!
- 10/24/2022. Hosung received the best elevator pitch award at the Department of Biological Chemistry Retreat!
- 11/16/2022. Wonsuk received the 2022 Samuel French Memorial Award!
- 12/18/2022. Alina, Sanghee, Nikki, and Varvara received UROP Fellowships!
- 3/1/2023. Johnny received the NIDDK F31 Fellowship!
- 5/1/2023. We received our first R21 grant!
- 6/20/2023. Grace and Johnny‘s paper is out in Nature Metabolism! Quantitative analysis of metabolic fluxes in brown fat and skeletal muscle during thermogenesis | Nature Metabolism
- 11/24/2023. Sunhee received an ADA postdoctoral fellowship!
- 1/25/2024. Johnny received the Behrens Research Excellence Award!
Funding Sources
We very much appreciate generous funding from the NIH, foundations, and research centers.
Contact:
Lab: 839 Health Sciences Road, Sprague Hall 150, UC Irvine, Irvine CA 92697
Office: 839 Health Sciences Road, Sprague Hall 122, UC Irvine, Irvine CA 92697.
Postdoc and student positions are available.
We are looking for lab members who are highly motivated to study organismal metabolism. Email your CV to choljang@uci.edu